Transcriptionnal regulation of RNA polymerase II at the level of the elongation phase
Transcription by RNA polymerase II can be divided in several stages, in particular the preinitiation, the initiation, the elongation and the termination. If the first two phases are clearly very important in the regulation of most genes, it was recently realized that the transition between initiation and elongation could be critical for modulation of the expression of about 30% of the genes trancribed by RNA polymerase II. Indeed, in many cases, the polymerase slows down and even stops completely (pauses) after synthesis of a few messenger RNA nucleotides. The switch of the enzyme into a processive elongation mode requires post-translational modifications of the largest subunit of RNA polymerase II. This subunit contains in its C-terminal domain many hexaptide repeats, which can be phosphorylated on serine 2 and 5. Serine 2 phosphorylation is particularly important to make the polymerase more efficient. This phosphorylation is carried out by a factor called p-TEFb composed of a kinase, Cdk9, and a cyclin co-factor, either cyclin T or cyclin K.
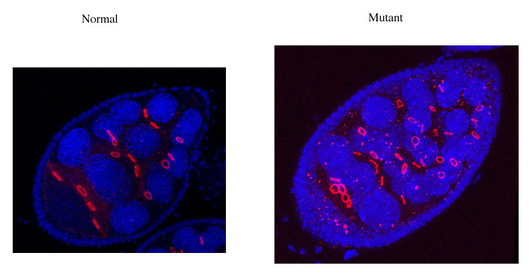
My laboratory uses genetic and molecular approaches in Drosophila melanogaster to analyse in vivo the function of p-TEFb complexes. We generated several transgenes that allow a conditional expression of an inactive Cdk9 kinase that competes with the normal kinase. This dominant negative approach allowed us to describe the effects of reducing p-TEFb activity in different tissues. We have described the importance of p-TEFb for cell proliferation and differentiation. It is also a step in the identification of genes whose expression is critically regulated at the level of the transition between initiation and elongation (release of RNA polymerase II pausing). We have also generated mutations and various transgenes for the two cyclins. We have shown that cyclin T and cyclin K are both essential and that the two types of p-TEFb complexes have different activities.